I Have a Crush on You!

Introduction
Severe crush injuries have the power to shatter lives in an instant. Imagine the sheer force of an external crushing weight bearing down on the human body, inflicting direct tissue trauma and triggering a cascade of devastating consequences. From muscle necrosis to neurologic dysfunction, the aftermath of a severe crush injury can be a race against time, where rapid identification and treatment by critical care and flight paramedics become the beacon of hope. In this article, we delve into the gripping world of crush injuries, unearthing the secrets to their prevention, the art of recognizing their subtle nuances, and the life-saving interventions you, as pre-hospital and critical care transport medicine providers, can employ. Brace yourself for a journey that reveals how swift action can alter the trajectory of these patients’ lives, transforming despair into new beginnings.
Definitions and Mechanism of Injury
Crush injuries are a specific type of trauma that occur when a body part or the entire body is subjected to a compressive force, often caused by heavy objects or structures. These injuries can result in severe tissue damage, disruption of vital functions, and potentially life-threatening complications. The injury mechanism involves applying prolonged or excessive pressure on the affected area, which compresses tissues, blood vessels, and nerves. This compression can impede blood flow, leading to ischemia and tissue death, while also causing the release of toxic byproducts into the bloodstream. Furthermore, crush injuries can accumulate metabolic waste products and inflammatory substances, further exacerbating tissue damage and potentially leading to systemic complications. The severity of crush injuries can vary widely, ranging from minor soft tissue injuries to complex fractures, compartment syndrome, and organ damage.

By IDF Spokesperson’s Unit, CC BY-SA 3.0, Link
Extrication and On-Scene Management
Crush injuries can result from numerous sources. While extrication is usually a great place to start, in large-scale mass trauma events, such as earthquakes or building collapses, where numerous victims are trapped, specialized teams called Urban Search and Rescue (USAR) have played a crucial role in successful rescue and medical assistance operations. These teams are comprised of various professionals, including EMTs and Paramedics, firefighters, law enforcement personnel, and government agencies, who work together to coordinate complex search and rescue efforts where the extrication might happen slowly over time.
One notable example of the effectiveness of USAR teams is the response to the devastating earthquake that struck Mexico City in 1985. The earthquake resulted in the collapse of numerous buildings, trapping thousands of individuals. USAR teams from different countries, including the United States, Israel, and Japan, were deployed to assist in the rescue efforts. Their expertise in confined space medicine allowed them to navigate through the rubble, providing medical care and extricating survivors. The coordination and collaboration among these teams played a pivotal role in saving lives during that catastrophic event.
The response to the 2011 earthquake and tsunami in Japan showcased the effectiveness of USAR teams in managing crush injuries. Japanese USAR teams and international assistance worked tirelessly to rescue victims trapped under the debris. Their expertise in confined space medicine and meticulous planning allowed for effective extrication and on-scene medical care. Their efforts saved numerous lives and demonstrated the critical role of USAR teams in managing crush injury incidents.
As a healthcare professional, you may already understand the significance of coordinated efforts between medical providers and rescue teams. Throughout this article, we will delve into the intricate details of managing crush injuries and explore scenarios where extrication may not always be the immediate priority. By fostering collaborative relationships and training alongside rescue teams, medical providers can enhance their understanding of rescue techniques while sharing valuable medical knowledge. This interprofessional collaboration ultimately improves patient outcomes and a more comprehensive approach to managing crush injuries.
Clinical Manifestations Resulting from Crush Injuries
Hyperkalemia
Hyperkalemia, an elevated level of potassium in the blood (serum K+ > 5.5 mEq/L), is a common and potentially life-threatening complication associated with crush injuries and rhabdomyolysis (we will talk more about this aspect in a minute). Crush injuries involving significant muscle damage can result in the release of intracellular potassium into the bloodstream, overwhelming the body’s ability to regulate potassium levels. The rapid onset of hyperkalemia can lead to cardiac arrhythmias, including ventricular fibrillation and cardiac arrest.
There are several EKG changes that you should be on the lookout for with hyperkalemia. You will see tall T waves (usually seen when K+ 5.0 – 6.0 mEq/L:
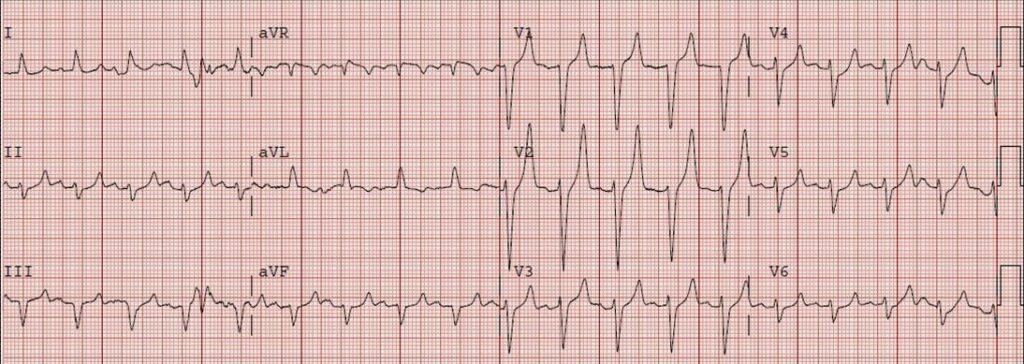
Flattened P waves and prolonged PR interval > 200 ms (usually seen when K+ 6.0 – 7.0 mEq/L):
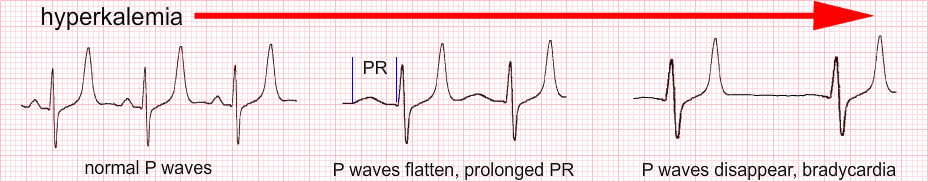
QRS waves > 0.12s (usually seen when K+ 7.0 – 8.0 mEq/L):
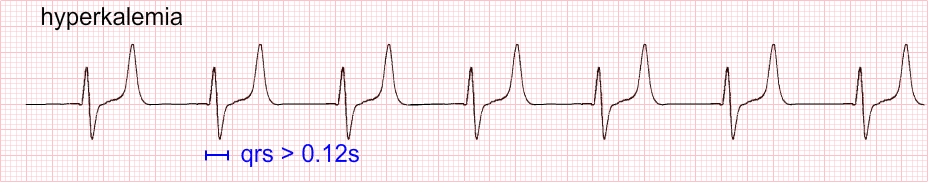
…and finally, if not corrected…
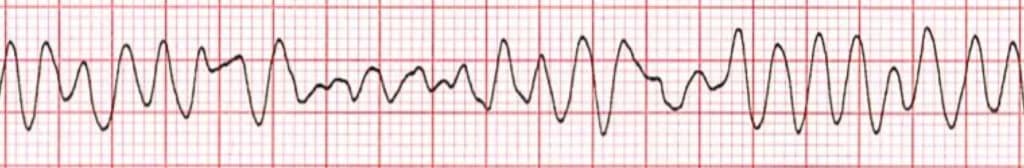

Prompt identification and management of hyperkalemia are crucial to prevent cardiac complications and improve patient outcomes, but without a “mobile laboratory,” how can you even identify hyperkalemia? Hyperkalemia is really only definitively diagnosed with lab values, right? The short answer is yes; however, certain clinical signs and symptoms may raise suspicion of hyperkalemia and prompt further evaluation.
In the context of crush injuries, providers should maintain a high index of suspicion for hyperkalemia due to the potential release of potassium from damaged muscle tissue. Clinical manifestations that may indicate hyperkalemia include muscle weakness, fatigue, palpitations, cardiac arrhythmias, and changes in electrocardiogram (ECG) readings. Specific ECG findings associated with hyperkalemia may include peaked T waves, prolonged PR interval, widened QRS complex, and eventually, sine wave pattern and ventricular fibrillation (as seen above).
While these clinical signs can raise suspicion of hyperkalemia, it is important to note that they are not definitive diagnostic criteria. Field identification of hyperkalemia is based on clinical suspicion, and definitive confirmation and management should be conducted in a medical facility where laboratory tests can accurately assess potassium levels and guide appropriate treatment strategies. In the meantime, let’s put the diagnosis of hyperkalemia secondary to crush injury up on our imaginary whiteboard of potential DDx, and continue doing what we do; ensuring a patent airway, administering high-concentration oxygen, establishing intravenous access, providing cardiac monitoring, and being good clinicians as we continue evaluating our patient. In terms of treatment for hyperkalemia secondary to crush injury, we can do some things that are beneficial.
Calcium gluconate:
Dosage: 1 gram of calcium gluconate (10 mL of a 10% solution) intravenously over 2-5 minutes. Administer slowly to reduce the risk of adverse reactions, such as bradycardia or hypotension. Repeat as necessary based on the patient’s response and electrocardiogram (ECG) changes. Calcium gluconate counteracts the effects of elevated potassium levels on the cardiac muscle. Hyperkalemia can lead to cardiac arrhythmias and cardiac arrest, and calcium gluconate is a stabilizer.
Sodium bicarbonate:
Dosage: 50-100 mEq of sodium bicarbonate intravenously as a slow infusion over 5-10 minutes. Administer as an adjunctive therapy in the presence of metabolic acidosis or severe hyperkalemia. Adjust the dosage based on the patient’s acid-base status and arterial blood gas analysis. Sodium bicarbonate works by increasing the pH of the blood, which promotes the movement of potassium ions from the extracellular fluid into the intracellular space. This shift is facilitated by the activity of the Na+/K+ ATPase pump, which transports potassium into the cells in exchange for sodium.
Inhaled beta-agonists (such as albuterol):
Dosage: 2.5-5 mg of albuterol via nebulizer every 20 minutes for up to three doses.
Administer via nebulization to facilitate bronchodilation and systemic beta-2 receptor stimulation. Monitor the patient’s response and adjust the frequency of administration as needed. By stimulating beta-2 receptors, these medications promote the cellular uptake of potassium, shifting it from the extracellular space into the intracellular compartment. This helps lower serum potassium levels and alleviate the effects of hyperkalemia.
Insulin with glucose:
Dosage: Intravenous regular insulin at a dose of 10 units, typically mixed with 25 grams of glucose as a 50 mL solution. Administer insulin and glucose as an intravenous infusion. Regularly monitor blood glucose levels and titrate the infusion rate to maintain euglycemia (normal blood sugar levels). Insulin stimulates the cellular uptake of glucose, which, in turn, promotes the uptake of potassium and glucose. This mechanism helps lower the potassium levels in the bloodstream.
Rhabdomyolysis
Rhabdomyolysis is a condition characterized by the breakdown of muscle tissue, leading to the release of myoglobin, creatine kinase, and other cellular components into the bloodstream. This can occur when prolonged compression or crush forces cause muscle necrosis and ischemia-reperfusion injury. If not promptly managed, the released myoglobin can overwhelm the kidneys’ filtration capacity, leading to acute kidney injury. Rhabdomyolysis can lead to electrolyte disturbances, such as hyperkalemia (as we have already discussed), hyperphosphatemia, and hypocalcemia.
Common clinical signs of rhabdomyolysis include muscle pain, tenderness, swelling, and weakness. Additionally, patients may present with dark-colored urine due to myoglobinuria. Laboratory investigations may reveal (if you could check them in the field) elevated serum creatine kinase levels, myoglobinuria, and electrolyte imbalances, including hyperkalemia and hyperphosphatemia.
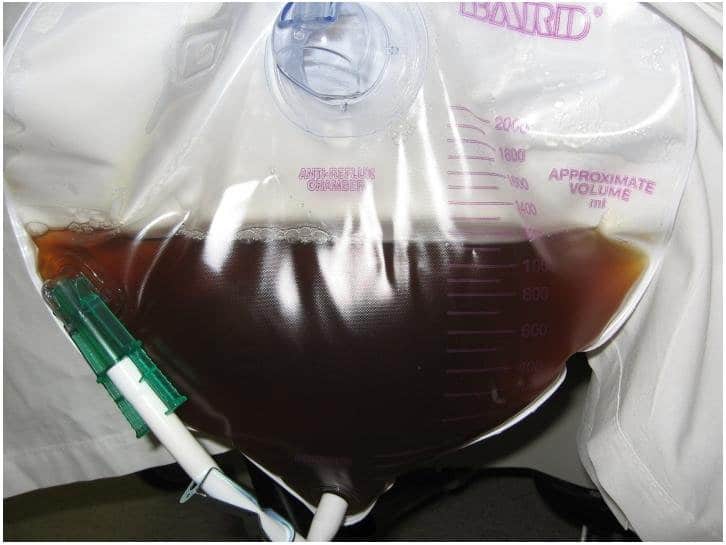
This is an example of the dark “tea-colored” urine we are taught about associated with rhabdomyolysis. It is a lot darker than you might think and should raise your index of suspicion that your patient may be in rhabdomyolysis.
Treating Rhabdomyolysis
Correction of rhabdomyolysis includes aggressive fluid hydration, which is crucial to maintain adequate renal perfusion and promote the flushing of myoglobin and other toxic substances from the kidneys. Intravenous fluids, such as isotonic saline, are typically administered at high volumes to achieve a urine output of 200-300 mL/hour (or approximately 3-5 mL/kg/hr in adults) or more.
The total fluid volume administered can range from 3-6 liters or more within the first 24 hours, depending on the patient’s condition. Initial bolus infusions of 1-2 liters of isotonic fluids may be administered rapidly to address hypovolemia and support renal perfusion. Following the bolus, a continuous infusion is initiated to maintain adequate hydration. The rate of infusion can vary but is often adjusted to achieve the desired urine output and maintain hemodynamic stability.
Alkalinization of urine can help prevent the formation of myoglobin casts and reduce the risk of acute tubular necrosis. This is achieved by administering bicarbonate or acetazolamide to raise urinary pH to a target range of 6.5-7.5. Alkalizing urine refers to the process of increasing the pH level of urine, making it more basic. Generally, the recommended oral dosage of sodium bicarbonate to alkalize urine ranges from 1 to 4 grams of sodium bicarbonate per day, divided into multiple doses.
As you can see, rhabdomyolysis is all centered around massive infusions of fluid, but as mentioned before, the guiding factors are the lab values. In the field, we often don’t see these on scene calls, but we do get the opportunity to review them during interfacility transports. Serial labs can give you a clue as to how the patient has responded prior to your transport. You are a fresh set of eyes for these patients, and reviewing patient charts should become an integral part of your interfacility transports. Not only can you help spot potential avenues for improving your patient, but you can also learn a lot from reading these charts. Let’s take a look at some tests that you will likely see in the chart of a patient with rhabdomyolysis:
Creatine Kinase (CK) Levels
CK is an enzyme found predominantly in skeletal muscles. Elevated CK levels in the blood are a hallmark of rhabdomyolysis. Serial measurements of CK levels are performed to monitor the extent of muscle injury and assess the response to treatment.
The normal range for CK levels can vary slightly depending on the laboratory and the specific reference range they use. However, as a general guideline, the normal range for CK in adults is typically around 30 to 200 international units per liter (IU/L) or 30 to 200 units per liter (U/L).
Serum Electrolytes
Tests for electrolyte levels, including potassium, calcium, phosphate, and magnesium, are important to monitor for imbalances that may arise due to muscle breakdown and renal involvement.
Here is a breakdown of the generally accepted normal ranges for serum electrolytes:
Sodium (Na+): 135-145 milliequivalents per liter (mEq/L)
Potassium (K+): 3.5-5.0 milliequivalents per liter (mEq/L)
Chloride (Cl-): 96-106 milliequivalents per liter (mEq/L)
Calcium (Ca2+): 8.5-10.5 milligrams per deciliter (mg/dL) or 2.1-2.6 millimoles per liter (mmol/L)
Magnesium (Mg2+): 1.7-2.2 milligrams per deciliter (mg/dL) or 0.7-0.9 millimoles per liter (mmol/L)
Renal Function Tests
Assessing renal function is crucial in rhabdomyolysis, as the condition can lead to acute kidney injury. Tests such as blood urea nitrogen (BUN) and creatinine are ordered to evaluate kidney function. Here are the normal ranges for some commonly measured renal function tests:
Blood Urea Nitrogen (BUN): 7-20 milligrams per deciliter (mg/dL) or 2.5-7.1 millimoles per liter (mmol/L).
Creatinine: 0.6-1.3 milligrams per deciliter (mg/dL) or 53-115 micromoles per liter (μmol/L) in adult males, and 0.5-1.1 milligrams per deciliter (mg/dL) or 44-97 micromoles per liter (μmol/L) in adult females.
Glomerular Filtration Rate (GFR): Normal GFR is typically greater than 90 milliliters per minute per 1.73 square meters (mL/min/1.73 m²).
Urinalysis
A urinalysis helps identify the presence of myoglobin, a protein released from damaged muscle cells, in the urine. The presence of myoglobinuria is a characteristic finding in rhabdomyolysis. Additionally, urinalysis can provide information about urine pH, specific gravity, and the presence of blood or casts.
Myoglobin in urine should be below a detectable range, or very low. Essentially, if myoglobin is detected at all in urine, it should be a big red flag that rhabdomyolysis is still happening.
Complete Blood Count (CBC)
A CBC is often performed to assess for any signs of infection or inflammation, monitor hemoglobin and hematocrit levels, and identify any associated complications. Let’s look at the normal ranges for CBC:
Hemoglobin (Hb):
Male: 13.5 to 17.5 grams per deciliter (g/dL)
Female: 12.0 to 15.5 g/dL
Hematocrit (Hct):
Male: 38.8% to 50.0%
Female: 34.9% to 44.5%
White Blood Cell count (WBC):
4,500 to 11,000 cells per microliter (c/μL) or 4.5 to 11.0 x 10^9/L
Platelet count (Plt):
150,000 to 450,000 c/μL or 150 to 450 x 10^9/L
Red Blood Cell count (RBC):
Male: 4.5 to 5.5 million c/μL or 4.5 to 5.5 x 10^12/L
Female: 4.0 to 5.0 million c/μL or 4.0 to 5.0 x 10^12/L
Liver Function Tests
Liver function tests may be ordered to assess liver enzymes and evaluate liver function, as certain causes of rhabdomyolysis can be associated with liver involvement. Here are the normal ranges for many common LFTs:
Alanine Aminotransferase (ALT):
Normal range: 7 to 55 units per liter (U/L)
Aspartate Aminotransferase (AST):
Normal range: 8 to 48 U/L
Alkaline Phosphatase (ALP):
Normal range: 40 to 129 U/L
Total Bilirubin:
Normal range: 0.1 to 1.2 milligrams per deciliter (mg/dL)
Albumin:
Normal range: 3.4 to 5.4 grams per deciliter (g/dL)
Total Protein:
Normal range: 6.0 to 8.3 g/dL
Gamma-Glutamyl Transferase (GGT):
Normal range: 9 to 48 U/L
Coagulation Profile
Coagulation studies, including prothrombin time (PT) and activated partial thromboplastin time (aPTT), may be conducted to evaluate the patient’s clotting status and identify any abnormalities. Here are the normal ranges for coagulation profile tests:
Prothrombin Time (PT):
Normal range: 11 to 13.5 seconds
International Normalized Ratio (INR):
Normal range: 0.8 to 1.2 (may vary based on specific medical conditions)
Partial Thromboplastin Time (PTT):
Normal range: 25 to 35 seconds
Platelet Count:
Normal range: 150,000 to 400,000 platelets per microliter (µL) of blood
Fibrinogen:
Normal range: 200 to 400 milligrams per deciliter (mg/dL)
D-dimer:
Normal range: Less than 0.5 micrograms per milliliter (µg/mL)
Arterial Blood Gas (ABG) Analysis
An ABG analysis helps assess acid-base balance and oxygenation status, particularly in patients with significant muscle injury or associated respiratory distress. Normal ABG ranges:
pH:
Normal range: 7.35 to 7.45
Partial Pressure of Oxygen (PaO2):
Normal range: 75 to 100 millimeters of mercury (mmHg)
Partial Pressure of Carbon Dioxide (PaCO2):
Normal range: 35 to 45 mmHg
Bicarbonate (HCO3-):
Normal range: 22 to 28 milliequivalents per liter (mEq/L)
Oxygen Saturation (SaO2):
Normal range: 95% to 100%
In almost all circumstances, there will be a printed report from the lab with all of these test results printed out in a way that is easy to read. However, you will commonly find test results in what are known as “fishbones.” These are a kind of shorthand for test results and are in the same pattern all the time so that even if they aren’t labeled, you can know what the values mean at a glance. Here are some common fishbones:

Continuous monitoring of renal function, electrolyte levels, and fluid balance is essential in severe cases of rhabdomyolysis. On the more severe end of the spectrum, renal replacement therapy, such as hemodialysis or continuous venovenous hemofiltration (CVVH), may be necessary to provide renal support and assist in removing myoglobin and other waste products.
Compartment Syndrome
Acute compartment syndrome can also occur as a result of crush injuries. It is characterized by increased pressure within a closed fascial compartment, leading to compromised tissue perfusion and potential nerve and muscle damage. Prolonged compression, swelling, and ischemia-reperfusion injury can contribute to the development of compartment syndrome. Common sites for compartment syndrome in crush injuries include the extremities, particularly the lower limbs. Compartment syndrome can occur for other reasons besides traumatic injury as well, including infection. For the purposes of this article, we are going to stick with injury, but know that there may be some overlap that you could apply in other cases.
Clinical manifestations of acute compartment syndrome include severe pain, progressive swelling, tense compartments, pallor, paresthesia, and loss of distal pulses. Early recognition and treatment of compartment syndrome are crucial to prevent irreversible tissue damage and limb loss. Compartment pressures can be measured using intracompartmental pressure monitoring devices, with a pressure reading of >30 mmHg being indicative of compartment syndrome (of course, these are not typical pieces of equipment in the pre-hospital setting, but are easy to learn and use).

There are no specific medications that can directly treat compartment syndrome since it is really more of a mechanical problem. Compartment syndrome is primarily managed through a prompt surgical intervention called fasciotomy, which involves making incisions in the affected compartments to relieve pressure and restore blood flow. This procedure is considered the gold standard treatment for compartment syndrome. Some analgesia would be greatly appreciated while the patient is in your care. Nonsteroidal anti-inflammatory drugs (NSAIDs) such as ibuprofen or naproxen are commonly used for mild to moderate pain relief. Opioid analgesics like morphine, oxycodone, or hydromorphone may be prescribed for severe pain that is not adequately controlled by NSAIDs alone. Most of these medications are not available to you in your pre-hospital drug kits, but do your best to control the pain with what you have within your protocols.
The following video has a lot of good information about compartment syndrome and the fasciotomy procedure that is used to correct the problem. The case in the video is a result of a medical cause of compartment syndrome rather than a traumatic one, but again the treatment is the same regardless of medical or traumatic origin. Check it out here: (you can also watch on YouTube directly by clicking here.)
Delaying Extrication
In some cases, immediate extrication of a crush injury victim may not be the first priority. Factors such as the stability of the rescue scene, the risk of further injury during extrication, and the presence of other life-threatening conditions can influence the order of interventions. For example, if a patient is trapped under debris in an unstable structure, unpinning them immediately may pose a significant risk of further injury or collapse. In such cases, stabilization of the rescue scene and ensuring the safety of the patient and rescuers become the primary focus. Additionally, some treatments may need to be initiated to prevent a cascade of toxins from negatively affecting the patient when they are freed.
Mortality
Mortality in severe crush injuries is often related to traumatic asphyxia, severe traumatic brain injury, and hemorrhagic shock. Crush syndrome related mortality varies based on patient factors, associated injuries, and the availability of resources. You play a HUGE role in reducing mortality with these patients. That’s what the Heroes on the Horizon campaign is all about! Getting some education into your hands to help improve patient outcomes involving traumatic injuries!
Thank you so much for reading! We hope this was educational and helps you make more informed decisions with your crush injury patients. As always, we want to encourage you to follow your local guidelines while providing care to your patients. Remember to keep learning, train together with other providers in your area, and communicate. This is how we reduce mortality and improve the tomorrows of all our patients.

Evan Claunch, BSB, FP-C, CCEMT-P, NRP
Director of Marketing and Information Technology at FlightBridgeED. With 21 years of experience as a paramedic in critical care transport medicine, I am proud to be an original co-founder of FlightBridgeED. I’m incredibly grateful for the support of our community over the past decade, and I’m excited for the future of FlightBridgeED. Together, we will continue to learn, collaborate, and innovate, ensuring that pre-hospital, air medical, and critical care transport medicine professionals have access to the best education and learning solutions available. Thank you for being part of our journey!

Disclaimer
The information provided in this article contains detailed descriptions of treatments, drug dosages, or other procedures. While these recommendations are based on generally accepted medical practice and research, it is important to note that medical practices can vary. Therefore, adhering to your local protocols, guidelines, and/or medical control orders is essential. The content in this article serves an educational purpose and should not replace medical practice guidelines or override the professional judgment of healthcare providers. FlightBridgeED assumes no responsibility or liability for actions taken by readers outside of their operating guidelines or local protocols. Always prioritize patient safety and individualized care based on each patient’s specific needs, following established medical guidelines.
